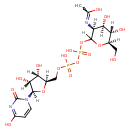
UDP-N-Acetyl-D-mannosamine (ECMDB20202) (M2MDB001048)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 2.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2012-05-31 14:32:54 -0600 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2015-09-17 15:41:49 -0600 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Secondary Accession Numbers |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | UDP-N-Acetyl-D-mannosamine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | UDP-N-acetyl-D-mannosamine is a member of the chemical class known as Pyrimidine Nucleotide Sugars. These are pyrimidine nucleotides bound to a saccharide derivative through the terminal phosphate group. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C17H27N3O17P2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight: | Average: 607.3537 Monoisotopic: 607.081569477 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | LFTYTUAZOPRMMI-IUPDHGCPSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C17H27N3O17P2/c1-6(22)18-10-13(26)11(24)7(4-21)35-16(10)36-39(31,32)37-38(29,30)33-5-8-12(25)14(27)15(34-8)20-3-2-9(23)19-17(20)28/h2-3,7-8,10-16,21,24-27H,4-5H2,1H3,(H,18,22)(H,29,30)(H,31,32)(H,19,23,28)/t7-,8-,10+,11-,12-,13-,14-,15-,16?/m0/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 26575-17-7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | N-[(3S,4R,5S,6R)-2-({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(4-hydroxy-2-oxo-1,2-dihydropyrimidin-1-yl)oxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)-4,5-dihydroxy-6-(hydroxymethyl)oxan-3-yl]ethanimidic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | N-[(3S,4R,5S,6R)-2-[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(4-hydroxy-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy(hydroxy)phosphoryl}oxy(hydroxy)phosphoryl)oxy]-4,5-dihydroxy-6-(hydroxymethyl)oxan-3-yl]ethanimidic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | CC(=O)N[C@@H]1[C@H](O)[C@@H](O)[C@H](CO)OC1OP(O)(=O)OP(O)(=O)OC[C@@H]1O[C@@H]([C@@H](O)[C@H]1O)N1C=CC(=O)NC1=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as pyrimidine nucleotide sugars. These are pyrimidine nucleotides bound to a saccharide derivative through the terminal phosphate group. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Nucleosides, nucleotides, and analogues | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Pyrimidine nucleotides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Pyrimidine nucleotide sugars | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Pyrimidine nucleotide sugars | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic heteromonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | Uridine diphosphate-N-acetylglucosamine <> UDP-N-Acetyl-D-mannosamine Water + 2 NAD + UDP-N-Acetyl-D-mannosamine <>3 Hydrogen ion +2 NADH + UDP-N-Acetyl-D-mannosaminouronate UDP-N-Acetyl-D-mannosamine + 2 NAD + Water <> UDP-N-Acetyl-D-mannosaminouronate +2 NADH +2 Hydrogen ion UDP-N-Acetyl-D-mannosamine + NAD + Water <> UDP-N-Acetyl-D-mannosaminouronate + NADH + Hydrogen ion Uridine diphosphate-N-acetylglucosamine > UDP-N-Acetyl-D-mannosamine UDP-N-Acetyl-D-mannosamine + 2 NAD + Water > UDP-N-Acetyl-D-mannosaminouronate +2 NADH Uridine diphosphate-N-acetylglucosamine <> UDP-N-acetyl-D-mannosamine + UDP-N-Acetyl-D-mannosamine UDP-N-acetyl-D-mannosamine + 2 NAD + Water + UDP-N-Acetyl-D-mannosamine > UDP-N-acetyl-α-D-mannosaminuronate +2 NADH +3 Hydrogen ion | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMPDB Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KEGG Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EcoCyc Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in UDP-N-acetylglucosamine 2-epimerase activity
- Specific function:
- Catalyzes the reversible epimerization at C-2 of UDP-N- acetylglucosamine (UDP-GlcNAc) and thereby provides bacteria with UDP-N-acetylmannosamine (UDP-ManNAc), the activated donor of ManNAc residues. Also involved in bacteriophage N4 adsorption
- Gene Name:
- wecB
- Uniprot ID:
- P27828
- Molecular weight:
- 42244
Reactions
| UDP-N-acetyl-D-glucosamine = UDP-N-acetyl-D-mannosamine. |
- General function:
- Involved in oxidation-reduction process
- Specific function:
- Catalyzes the four-electron oxidation of UDP-N-acetyl-D- mannosamine (UDP-ManNAc), reducing NAD(+) and releasing UDP-N- acetylmannosaminuronic acid (UDP-ManNAcA)
- Gene Name:
- wecC
- Uniprot ID:
- P27829
- Molecular weight:
- 45838
Reactions
| UDP-N-acetyl-D-mannosamine + 2 NAD(+) + H(2)O = UDP-N-acetyl-D-mannosaminuronate + 2 NADH. |